Cardiac and Vascular Imaging
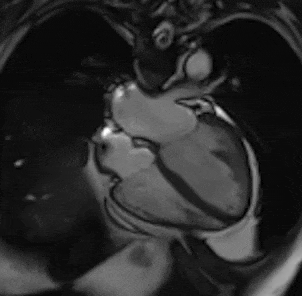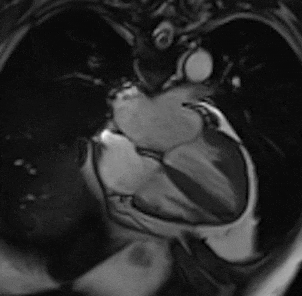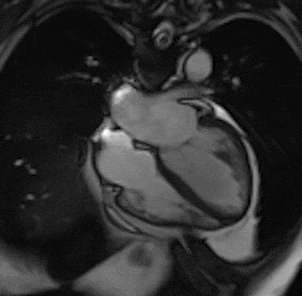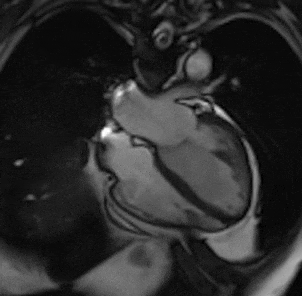
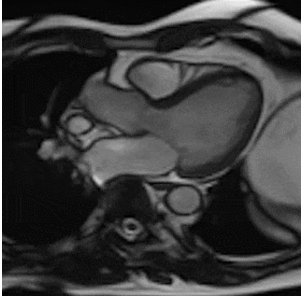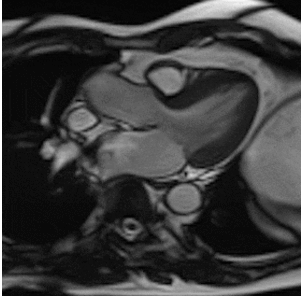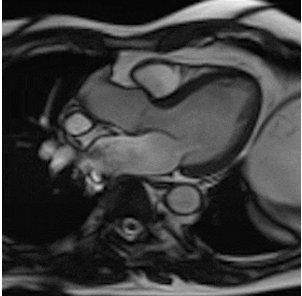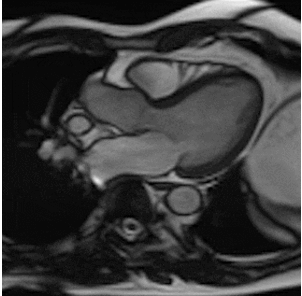
By using different pulse sequences we can help visualize:
-
evaluating the anatomy and function of the heart chambers, valves, size and blood flow through major vessels and surrounding structures such as the pericardium (the fluid sac that surrounds the heart).
-
diagnosing a variety of cardiovascular (heart and/or blood vessel) disorders such as tumors, infections and inflammatory conditions.
-
evaluating the effects of coronary artery disease such as limited blood flow to the heart muscle and scarring within the heart muscle after a heart attack.
-
evaluating the effects of surgical changes, especially in patients with congenital heart disease.
- Blood flow through the cavities of the heart and great vessels is pulsatile and is subject to time and multidirectional variations. To date, the recording of blood flow in multiple directions and phases has been limited. 4D-flow MRI offers advantages for the recording, visualization and analysis of blood flow.
CFMM Collaborative Studies
Miracle
PI: DR Sposato, Luciano
Department: Robarts Research
Worldwide, stroke causes 1 million cardiovascular deaths per year and a similar number of non-fatal post-stroke major adverse cardiovascular events. In Canada, 9.1% of individuals without previous heart disease experienced these adverse events after a year after their stroke. The risk for these adverse events was highest within the first month post-stroke, suggesting a strong link between both conditions and highlighting an important missed opportunity for prevention. These adverse cardiovascular events are thought to be caused by inflammation. Interleukin-1 (IL-1) is produced in the brain post-stroke and plays a central role in this causation. Once in the bloodstream, it stimulates the production of catecholamines by the adrenal gland and enhances the activation/release of neutrophils and macrophages in the spleen and bone marrow, further promoting IL-1 synthesis. In the heart, IL-1 causes cardiac inflammation, which in turn causes endothelial dysfunction, coronary plaque rupture, impaired myocardial contractility, and myofibroblast activation causing subendocardial fibrosis ultimately triggering cardiac arrhythmias. The mechanism by which post-stroke inflammation causes heart disease is known as stroke-induced heart injury (SIHI). The present study is an investigator initiated, single-centre, prospective cohort study. The study aims to enroll 10 patients with acute/subacute middle cerebral artery (MCA) ischemic stroke. We will compare inflammatory markers among 44 patients with MCA ischemic stroke within 10 days post-stroke vs. 60±15 days post-stroke, and we will measure B-Type Natriuretic Peptide at 10 and 60±15 days post-stroke. Participants with elevated troponin T levels will undergo MR scanning of the heart within 10 days and 60±15 after stroke onset, in order to measure the degree of cardiac inflammation. We estimate 25% of patients will satisfy this condition (n=11)..
MAPIT-CRT
PI: Drangova, Maria
Department: Robarts Research
Cardiac Resynchronization Therapy (CRT) is a permanent pacing therapy aimed at re-timing the contraction of the right and left ventricular (RV & LV) walls to improve the hearts pumping function. Even though CRT reduces hospitalization, improves symptoms and reduces mortality, over 40% of patients do not seem to respond due to the presence of scar and improper positioning of RV and LV leads. The purpose of the MAPIT-CRT study is to identify if using an MRI to guide and target the optimal location of the LV and RV pacing leads will improve the hearts response to CRT. MAPIT-CRT is a double blinded, randomized clinical trial being performed at 5 centres across Canada. Once enrolled patients will be followed for a year to evaluate their response to CRT.
START
PI: Drangova, Maria
Department: Robarts Research
Patients who have suffered heart attacks or who have genetic or inflammatory heart disease frequently have scarred heart muscle. This scarring is a barrier to improvement by medical therapy and promotes sudden cardiac death (SCD) due to arrhythmia. Scar imaging may allow for more optimal delivery of specialized pacemaker leads that are designed to both improve heart function and shock the heart out of life threatening heart rhythms. BIRC investigators are studying MRI scar imaging to identify patients likely to suffer SCD, using Gd-DTPA as a contrast agent to precisely delineate the anatomic extent and characteristics of this scar. This is applicable to a variety of cardiac diseases including myocardial infarction and a number of inflammatory conditions. This may play a major role in guiding clinicians to make management decisions to improve patient outcomes.
Aortic Microstructure assessed with DT-MRI
PI:Pickering, Geoffery
Department: Medical Biophysics
This project addresses the critical health problem of thoracic aortic dissection and rupture. The thoracic aorta receives blood ejected from the heart and is biomechanically structured to withstand the unrelenting forces. However, mechanical failure can occur and can be catastrophic for at-risk individuals. This includes individuals with genetic aortopathies (eg. Marfan disease), those born with a bicuspid aortic valve (2% of the population), and older individuals (degenerative aortopathy). The primary approach to lessening the life threatening risks of aortic wall tearing and rupture is aortic diameter surveillance and prophylactic surgical replacement of part of the thoracic aorta, once a threshold diameter is reached. However, the guidelines are population-based and relatively crude. In fact, the recommended threshold diameters may fail to capture up to 50% of those that will sustain an aortic dissection. Personalized strategies are required.
Currently, computed tomography, echocardiography, and magnetic resonance imaging are widely used strategies to ascertain aortic diameter. Missing from these assessments is information on the composition and archirtecture of the aortic wall itself. This is an important gap because aortic vulnerability is defined by structural degeneration of the aortic media. Hallmark features are elastin breakdown, proteoglycan accumulation, and smooth muscle cell loss and dissarray. Non-invasive imaging tools for delineating aortic wall microstructure could provide vital, patient-specific information on aortic vulnerability. However, no such tools exist.
Using high-field MRI, we have discovered that diffusion tensor MRI can reveal the ordered cyto-architecture of the normal, porcine thoracic aorta. Furthermore, we have established that tractography, fractional anisopropy, and mean diffusivity can identify enzymatically dusrupted regions of the porcine aortic wall. We have also successfully undertaken ex vivo CEST-MRI imaging in normal and dilated human thoracic aortas and delineated regions of structural disruption and proteoglycan accumulation. We are now in a position to build on these exciting findings to develop protocols to image the human thoracic aorta in vivo with DT-MRI. Our current data, recent advances in cardiac and respiratory gating, and existing sequences on the scanners make this an exciting and feasible study, and hopefully the first use of 3D tractography in human aortas.





